Understanding cells as interactive societies during embryonic development
Cell Competition
During early embryonic development, cells undergo highly coordinated programs that shape a uniform mass of cells into complex and functional bodies. However, making these cell fate choices and withstanding the epigenetic, mechanic, and metabolic rewiring, while simultaneously proliferating at record speed, does not come without complications:
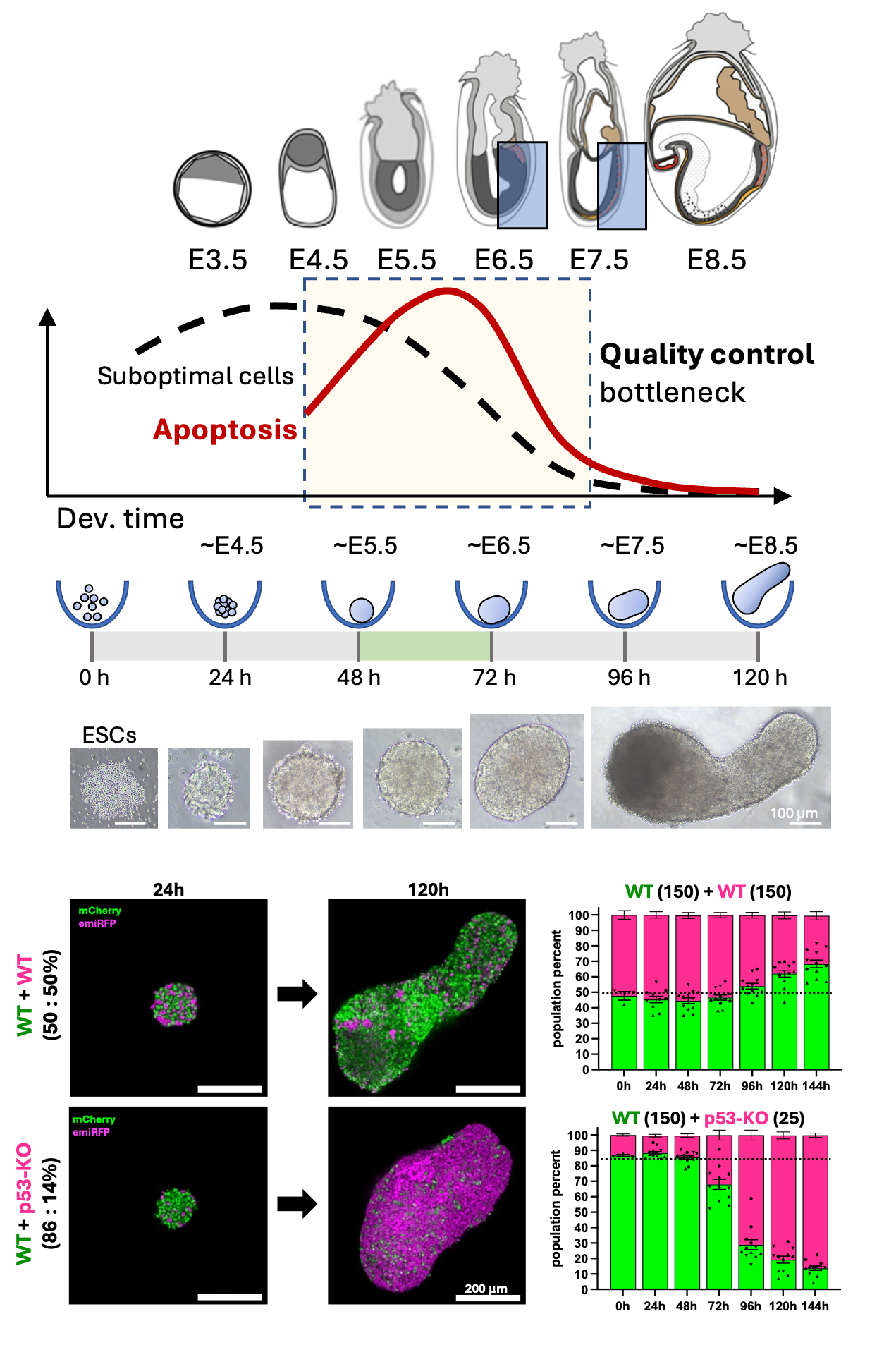
~70% of human pre-implantation embryos contain aneuploid cells and around 30% of conceptions are lost in the first 3 weeks. To counterbalance this, embryos use stringent quality controls to make sure that only the fittest cells contribute to the final organism, while suboptimal cells are removed.
In mouse embryos, right before gastrulation (E5.5-E6.5), cells undergo a non-autonomous quality control mechanism called Cell Competition, which coincides with a wave of cell death in the epiblast. During cell competition, cells compare their own fitness to that of their neighbors, after which cells that are viable but less fit than their surrounding undergo apoptosis. Interestingly, even healthy cells undergo apoptosis when in proximity to so called ‘supercompetitor’ cells, which can be defined by lack of tumorsuppresssor p53, or over-abundance of oncogene cMyc.
The central questions of our group are: how do cells communicate with each other during this process, how do they make their life and death decisions as a result of this communication, and how is all this regulated in space and time during 3D embryonic development.
Research focus
Gastruloids
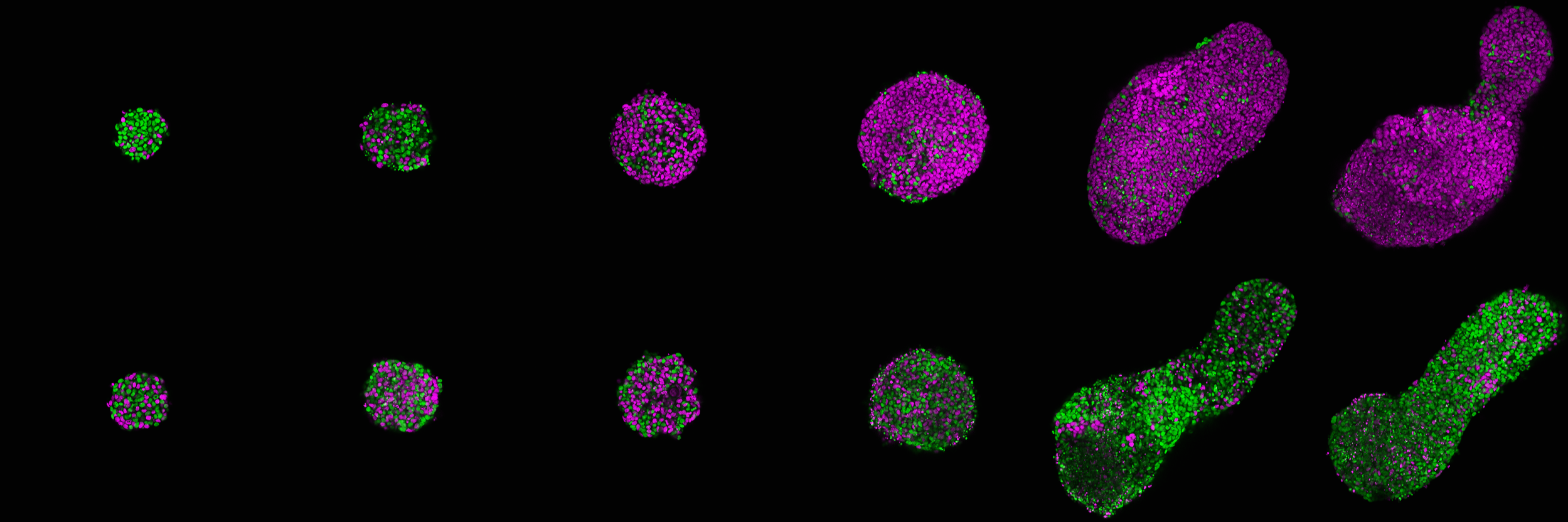
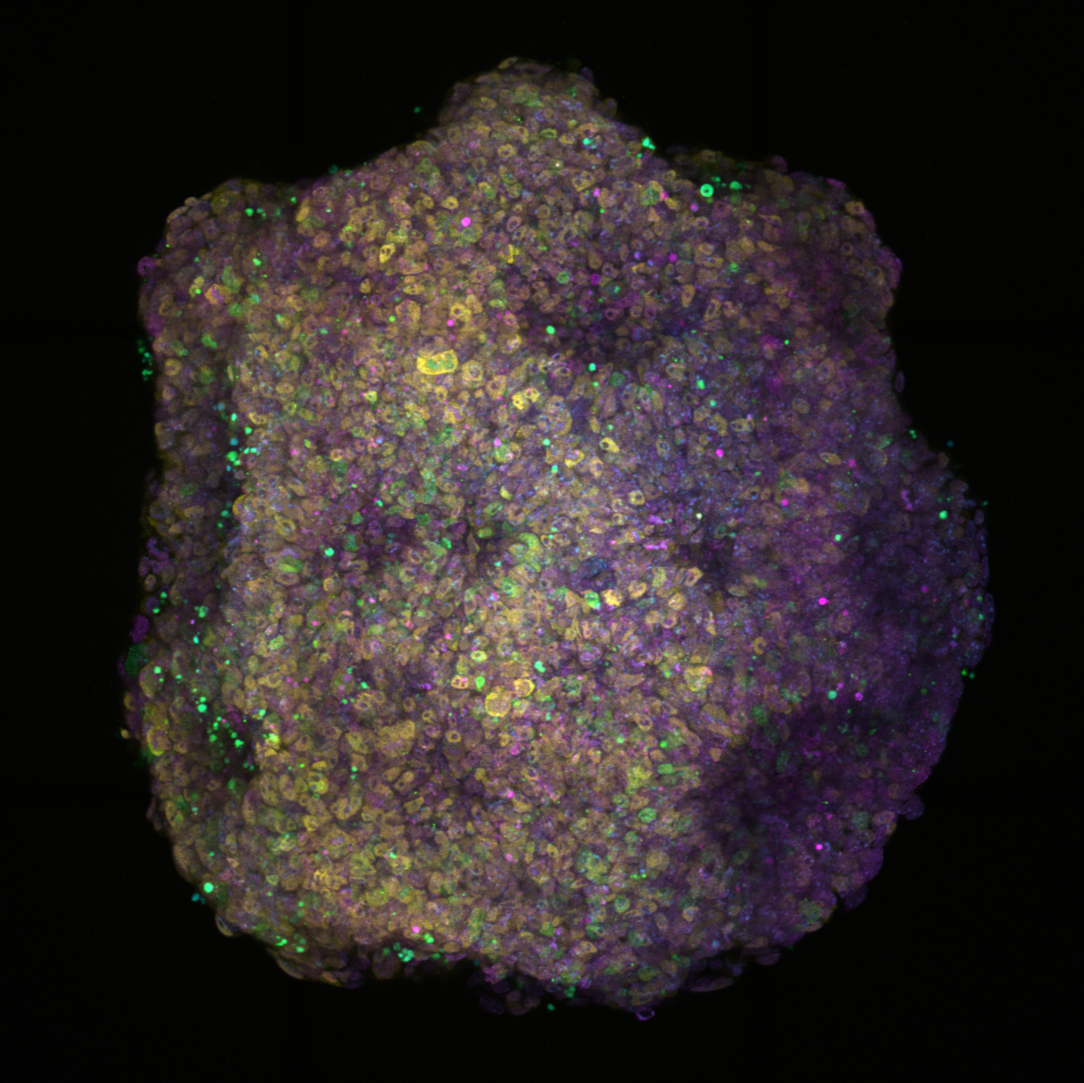
To study cell competition in a 3D developmental system, we use gastruloids, an embryonic stem cell-based in vitro system that models the core processes of embryonic development from naive pluripotency, through gastrulation, up to somitogenesis in a spatio-temporally coordinated manner.
By generating mosaic gastruloids containing cells of varying fitness levels, we obtain a dynamic model of defined cell-cell interactions that simultaneously offers full perturbation, live imaging, as well as high-throughput screening. Using degron systems, we additionally gain dynamic control of cellular fitness machineries and can create “on-demand supercompetitors”.
While the foundation of our work is based on mouse gastruloids, we are expanding into a new version of human gastruloids, as well as gastruloids of non-model species, particularly those with enhanced intrinsic resistance to cancer and altered quality control machineries.
Cell-Cell Communication
One of the central questions in the cell competition field remains: “How do cells communicate their fitness to each other?”. The biological system we use create an optimal 3D setup to tackle exactly that question from all fronts at once, including high-throughout CRISPR and pharmacolocial screens, proteomics, and and time-resolves transcriptomics.
Cell intrinsic vs non-autonomous quality control in cancer resistant species
Evolution has gifted us multiple species with phenomenal cancer resistance. Examples of these are Naked Mole Rats, which live for up to 30 years, and Elephants, which not only have very long life-spans, but also undergo countless cell divisions to establish their large body mass. Both these species almost never develop tumors. While this is attributed to different species-specific aspects (like the 20 copies of TP53 in the elephant genome together with a unique LIF6 stress response, and hypersensitive p53 and Rb stress-response in naked mole rats), the common ground in both is a heightened cell-intrinsic stress response. By using stem cell based developmental in vitro systems and iPSCs of these species, we ask the question how their altered intrinsic machineries affect cell non-autonomous competition during development. Has evolution provided these species with such rigorous intrinsic safety nets that they no longer have use for non-autonomous cell competition? Or are these species excellent competitors due to their heightened sensitivity? Does this excellent cell competition in turn contribute to their cancer resistance? Let’s find out!